 .
.
Juan José Serrano Silva, MD1, Sandra Fernández, Biol. MSc2, Nicolás Rosillo, MD, PhD3, Héctor Bueno, MD, PhD4
doi: http://dx.doi.org/10.5195/jmla.2026.2262
Volume 114, Number 1: 83-85
ABSTRACT
Covidence. Covidence Pty Ltd, Level 10, 446 Collins ST, Melbourne VIC 3000, Australia; support@covidence.org; https://www.covidence.org/; pay per review.
Rayyan. Rayyan, 1 Broadway, 14th Floor Cambridge, MA, 02142 USA; https://www.rayyan.ai/; pay per user.
EPPI Centre. EPPI Centre, Social Science Research Unit, UCL Social Research Institute, 10 Woburn Square, London WC1H 0NS; eppisupport@ucl.ac.uk; https://eppi.ioe.ac.uk/cms/; pay per user.
Distiller SR. DistillerSR Inc, 505 March Road, Suite 450, Ottawa, Ontario, Canada, K2K 3A4; support@distillersr.com; https://www.distillersr.com/; contact for pricing.
RevMan. The Cochrane Collaboration, 11-13 Cavendish Square, London, W1G 0AN, United Kingdom; https://revman.cochrane.org/info; pay per user.
Systematic reviews are critical in evidence-based medicine, yet their execution demands substantial resources in both time and personnel. The growing volume of scientific publications, the adoption of increasingly rigorous methodological standards, such as PRISMA [1,2], the use of evidence-quality assessment tools [3] and the need of conducting exhaustive searches across multiple databases [4] have amplified their complexity and workload. This complexity underscores the need for specialized tools to optimize the review process. This analysis summarizes and compares the leading software for systematic reviews and meta-analyses, showing how an informed choice can enhance both efficiency and quality. To this end, we conducted a targeted literature review of the most commonly used software for systematic reviews and meta-analyses followed by a critical evaluation of their features to guide researchers in selecting the tool best suited to their needs.
The most widely used softwares for conducting systematic reviews are Covidence [5], Rayyan [6], EPPI-Reviewer [7], DistillerSR [8], and Review Manager (RevMan) [9].
Covidence is widely recognized for its intuitive interface—usually associated with a shorter learning curve— and its capacity to streamline screening and data extraction. As a web-based platform, it facilitates real time collaboration among team members. Its pricing model is based on a per-review fee, allowing unlimited users per project, an advantage for teams with many contributors. Rayyan, in contrast, offers a basic free version (with optional paid) and leverages artificial intelligence to accelerate screening and duplicate detection. It is particularly accessible and integrates well with reference managers. Its paid model is user-based, making it potentially more cost-effective for smaller teams. However, Rayyan lacks built-in functionalities for data extraction and quality assessment, which limits its utility beyond the initial screening phases. Despite these limitations, both Covidence and Rayyan are excellent, low-cost solutions for researchers prioritizing efficiency and collaboration in the early stages of a systematic review. Neither, however, offers meta-analysis capabilities.
For more advanced requirements, platforms such as EPPI-Reviewer or DistillerSR provide extended functionalities, including machine learning tools and comprehensive process automation. These solutions offer greater power and flexibility but are generally more complex, with steeper learning curves and higher costs. Their ability to integrate with other systems and workflows varies by platform. RevMan, the reference software supported by the Cochrane Foundation, stands out for its user-friendly environment for data analysis and writing. Although it lacks automation capabilities and robust screening functionalities, it includes built-in meta-analysis functions and generates standard graphs such as forest plots. Its limited interoperability with external applications, however, may be a constraint in more integrated or customized workflows. A detailed comparison of the features, strengths, and limitations of these platforms is provided in Table 1.
Table 1A comparative analysis of the features, strengths, and limitations of leading software for systematic reviews.
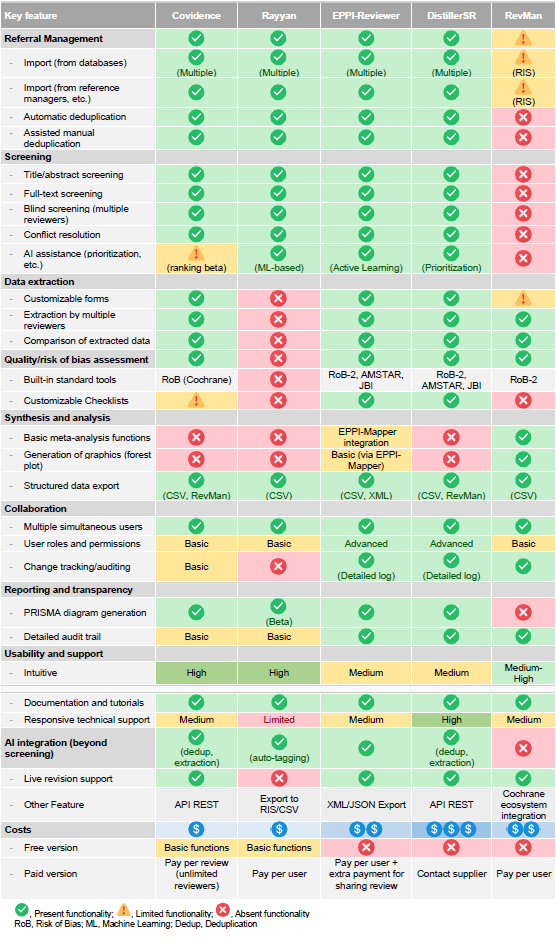
While it is theoretically possible to conduct a systematic review without dedicated software, doing so is inefficient, time-consuming, and increases the risk of error. Critical stages such as duplicate removal, study screening and quality assessment, data extraction, and collaborative analysis can benefit substantially from the use of specialized tools. The selection of a specific platform depends on multiple factors, including the complexity of the review, team size, budget constraints, required functionalities, acceptable learning curve, and compatibility with the researcher's existing digital ecosystem.
Importantly, the choice of software does not have to be limited to a single tool. An optimal workflow may involve the combined use of several platforms—such as employing Rayyan for its efficient screening capabilities, followed by export to RevMan for meta-analysis and reporting. Therefore, prioritizing and tailoring tool selection to specific needs of each phase is essential. Ultimately, the strategic use of appropriate software is critical to enhancing the efficiency of research teams and ensuring the methodological rigor and overall quality of systematic reviews.
The authors declare no conflicts of interest related to the content of this letter.
1. Krnic Martinic M, Pieper D, Glatt A, Puljak L. Definition of a systematic review used in overviews of systematic reviews, meta-epidemiological
studies and textbooks. BMC Med Res Methodol. 2019 Dec;19(1):203. http://dx.doi.org/10.1186/s12874-019-0855-00855-0 .
.
2. Johnson BT, Hennessy EA. Systematic reviews and meta-analyses in the health sciences: Best practice methods
for research syntheses. Soc Sci Med. 2019 Jul;233:237–51. http://dx.doi.org/10.1016/j.socscimed.2019.05.035 .
.
3. Shehata N, D’Souza R. Conducting a High-Quality Systematic Review. J Rheumatol. 2025 Apr;jrheum.2024-1241. http://dx.doi.org/10.3899/jrheum.2024-1241.C11241.C1 .
.
4. Bramer WM, Rethlefsen ML, Kleijnen J, Franco OH. Optimal database combinations for literature searches in systematic reviews: a prospective
exploratory study. Syst Rev. 2017 Dec;6(1):245. http://dx.doi.org/10.1186/s13643-017-0644-y0644-y .
.
5. Covidence Pty Ltd. Covidence: the world’s #1 systematic review tool [Internet]. Melbourne, Australia [cited 26 jun 2025]. https://www.covidence.org/https://www.covidence.org/.
6. Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016 Dec;5(1):210. http://dx.doi.org/10.1186/s13643-016-0384-40384-4 .
.
7. EPPI Centre. EPPI Centre, evidence for Policy & Practice [Internet]. London, United Kingdom: Social Science Research Unit, UCL Institute of Education [cited 26 jun 2025]. https://eppi.ioe.ac.uk/cms/.
8. Distiller SR Inc. DistillerSR. Smarter reviews: trusted evidence [Internet]. Ontario, Canada [cited 26 jun 2025]. https://www.distillersr.com/.
9. The Cochrane Collaboration. RevMan: systematic review and meta-analysis tool for researchers worldwide [Internet]. United Kingdom [cited 26 jun 2025]. https://revman.cochrane.org/info.
Juan José Serrano Silva, MD, 1 juanjo.serrano.silva@gmail.com, Cardiology Department, Hospital Universitario de Jerez de la Frontera, Jerez de la Frontera, Spain; Centro Nacional de Investigaciones Cardiovasculares (CNIC), Madrid, Spain
Sandra Fernández, Biol. MSc, 2 sandra.fernandez@cnic.es, Centro Nacional de Investigaciones Cardiovasculares (CNIC), Madrid, Spain; Instituto de Investigación Sanitaria Hospital 12 de Octubre (i+12), Madrid, Spain
Nicolás Rosillo, MD, PhD, 3 nicorora93@gmail.com, Cardiology Department, Hospital Universitario 12 de Octubre, Madrid, Spain; Instituto de Investigación Sanitaria Hospital 12 de Octubre (i+12), Madrid, Spain
Héctor Bueno, MD, PhD, 4 hbueno@cnic.es, Centro Nacional de Investigaciones Cardiovasculares (CNIC), Madrid, Spain; Cardiology Department. Hospital Universitario 12 de Octubre, Madrid, Spain; Instituto de Investigación Sanitaria Hospital 12 de Octubre (i+12), Madrid, Spain
© 2026 Juan José Serrano Silva, Sandra Fernández, Nicolás Rosillo, Héctor Bueno
This work is licensed under a Creative Commons Attribution 4.0 International License.
Journal of the Medical Library Association, VOLUME 114, NUMBER 1, January 2026